LDPE (Low Density Polyethylene)
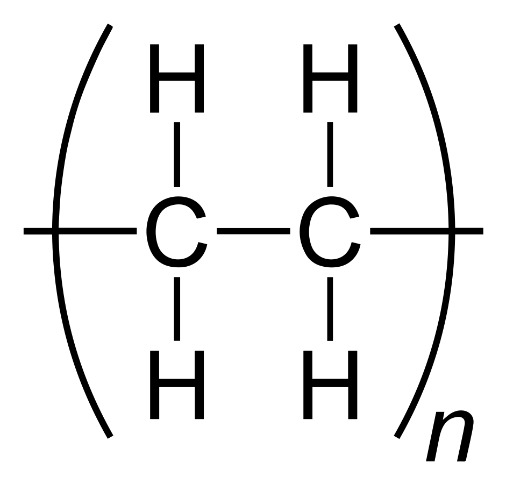
Polyethylene is a thermoplastic polymer belonging to the Polyolefins family. It is obtained from the polymerization of ethylene and is one of the most used and processed polymers, constituting the largest proportion of polymers worldwide. Its technical characteristics strongly depend on its molecular weight, crystallinity rate and molecular weight distribution. Polyethylene is used in different densities: low (LDPE), medium (MDPE) and high (HDPE).
Chemical Formula LDPE: (C2H4)n
CAS number PE: 9002-88-4
PE (IUPAC) Chemical Number: low-density polyethylene
Recycling Code PE: numero 4
Recycling Symbol PE: 
LDPE commercial names and producers:
- Borpure (Borealis)
- Stamylan (Sabic)
- Alcudia (Repsol)
- Attane (DOW)
- Borealis PE (Borealis)
- Borstar (Borealis)
- Clearflex (Versalis)
- Eltex (Ineos Polyolefines)
- Eraclene (Versalis)
- Escorene (Exxon Mobil)
- Alastian (LyondellBasell)
- Lucalen (LyondellBasell)
- Lupolen (LyondellBasell)
- Petrothene (LyondellBasell)
- Purell (LyondellBasell)
- SURPASS (NovaChem)
- Braskem PE (Braskem)
- Total PE (Total Petrolchemicals)
LDPE Italian distributors:
- BIGLER (Total LDPE)
- Schulman (Network polymers, Accuguard, Accucomp)
- Mega Polymers (LUPOLEN, STAMYLAN)
- PolyOne (AT LDPE, DOW Endurance, DOW LDPE, DuPont 20 series, Marlex, Maxxam, Westlake)
- ALBIS (Lucalen, Lupolen, Malen, Purell)
- Interpolimeri (ExxonMobil LDPE)
- Biesterfield (Eltex, Novex)
- Nexeo (Bormed, Braskem, Bynel, DuPont 20 series, HIVAL, Microthene, NOVAPOL, Petrothene, Purell, SABIC LDPE)
- RESINEX (DOW Endurance, DOW LDPE, Ravalene)
- Ultrapolymers (LUVOCOM, Lucalen, Lupolen, Purell, Ravalene)
- TER Plastics (Riblene)
Main compounds LDPE:
- LDPE with antistatic agent
- LDPE with UV stabilizers
Chemical and Physical properties
LDPE is a semi-crystalline material. Its level of crystallinity is limited due to the decay of the chains. It is a resistant material, but has moderate elasticity and tends to permanently deform. It is however characterized by a good level of resilience and chemical resistance. The material rolls easily due to the decay of long chains.
The materials on the market have densities between 0.91 and 0.94 g / cm³. LDPE is permeable to gases and vapors. It is resistant to many solvents at room temperature, but aromatic and chlorinated hydrocarbons cause it to swell. It has an excellent impermeability to water and for this reason no drying treatments are necessary.
LDPE standard density: 0,92 g/cm3 (ISO 1183)
LDPE melting point Tm: 110 °C
LDPE glass transition temperature Tg: -130 °C
Density differences between the main types of PE::
LDPE: density range from 0,91 g/cm to 0,925 g/cm
MDPE: density range from 0,926 g/cm to 0,94 g/cm
HDPE : density range from 0,94 g/cm to 0,965 g/cm
LDPE market applications
- Lids
- Bowls
- Containers
- Barrier film for foods
- Coatings
- Construction
- Disposable products
LDPE transformation technologies
- Injection molding
- Blow molding
- Extrusion
History and Trivia on Polyethelene:
Polyethylene or Polythene is one of the oldest polymers and still the most used today. Discovered almost accidentally by E.W. Fawcett and R. O. Gibson, its first production came about on September 1, 1939, on the same day that World War II began. Initially it was only considered to be a material with excellent insulating properties against electricity. During the Second World War it was used in large quantities in radar-related applications. At the end of the war it was in danger of disappearing until its properties were discovered to be much more versatile than previously thought. Fundamental in the world of plastic was the discovery by Karl Ziegler of the complete dimerization reaction of ethylene in butylene. Immediately afterwards he discovered the way to obtain high linear ethylene polymers under low temperature and pressure conditions. These discoveries marked the beginning of the revolution in the world of polymers.
Disclaimer: Il contenuto di questo database è stato creato sulla base di informazioni di pubblico dominio ed è gestito da PlasticFinder srl. Le descrizioni contenute non offrono alcuna garanzia di completezza, attualità o accuratezza. Eventuali commenti o integrazioni possono essere inviate all'indirizzo e-mail: info@plasticfinder.it
PlasticFinder s.r.l. non verifica che gli utenti abbiano il diritto di disporre dei prodotti descritti nella scheda-prodotto, né la titolarità dei diritti di proprietà intellettuale e non è responsabile di eventuali violazioni di diritti di terzi.
| LDPE First choice - Virgin | FLEXIRENE CL 10 S | granule/pellet | Natural / Neutral | 23375 Kg | bigbag 1.000-1.100 kg | 1.09 €/Kg | |
| LDPE Recycled SRM /Regrinded / Industrial By-Product / Industrial compound | RPE NERO-LDPE RICICLATO NERO MISC.UNI10667-2 | granule/pellet | Black | 55000 Kg | bigbag 1.000-1.100 kg | 0.84 €/Kg | |
| LDPE Recycled SRM /Regrinded / Industrial By-Product / Industrial compound | - | granule/pellet | Miscellaneous | 600 Kg | octabin | 1.00 €/Kg | |
| Polymer LDPE FLEXIRENE CL 10 S | 23375 Kg | 1.09 €/Kg | |
| Polymer LDPE RPE NERO-LDPE RICICLATO NERO MISC.UNI10667-2 | 55000 Kg | 0.84 €/Kg | |
| Polymer LDPE | 600 Kg | 1.00 €/Kg | |
Polymer List
Polimeri attualmente disponibili su PlasticFinder Wiki:
- ABS (Acrilonitrile Butadiene Stirene)
- Ecoallene
- HDPE (Polietilene ad alta densità)
- LDPE (Polietilene a bassa densità)
- PA6 (Poliammide 6, nylon 6)
- PA66 (Poliammide 66, nylon 6-6, nylon 6/6 o nylon 6,6)
- PA12 (Poliammide 12, nylon 12)
- PBT (Polibutilene Tereftalato)
- PC (Policarbonato)
- PET (Polietilen Tereftalato)
- PMMA (Polimetilmetacrilato)
- POM (Polimetilmetacrilato)
- PP (Polipropilene - Polipropene)
- PVC (Cloruro di Polivinile)



